
Hi, I’m Marie Gale.
For over 15 years I’ve been researching and following the world of laws, regulations, standards and guidelines for the handcrafted soap and cosmetic industry. What started as a personal quest to understand soap and cosmetic labeling for my own products has grown into a deep understanding of the myriad of requirements that handcrafted soap and cosmetics makers are subject to.
My personal passion to assist others to understand and follow the labeling and other requirements grew out of all that research. If I had my way, no handcrafter would ever experience the stress and heartache of unwittingly running afoul of the the laws and regulations.
There is peace of mind in knowing that you are compliant with the applicable regulations. I’m here to help you achieve that stress-free relationship with your products and your business.
Whether you know it or not, you’ve been an invaluable resource for me throughout our years in business as I’ve relied on your many books whenever I have questions. I’ve recently purchased “Navigating the Rules & Regulations” and am going through it with tabs & a highlighter and am learning more than I ever expected. — Julia
Latest blog articles
-

Alcohol in Cosmetics
Ethyl, denatured, or isopropyl — what’s really in your product? Not all alcohols are the same, and they have different regulations and labeling requirements.
-

Safety Substantiation: What it Means (and How To Do It)
Before MoCRA, a cosmetic product was supposed to be safe, but there was no requirement for proof of its safety. Now proof is required.
-

Using the Open Jar and Hourglass Symbols
The open jar and hourglass symbols appear on many cosmetic labels—but in the U.S., they’re often misunderstood or misused. Here’s what they actually mean.
-

Glass vs. Plastic: Which Wins?
Choosing between glass and plastic isn’t just about looks. Explore the real trade-offs in sustainability, cost, safety, and perception — and find what fits your products best.
-

Incidental Ingredients: Declare or Not?
Incidental ingredients are trace materials with no function in a cosmetic. Here’s when you can omit them—and why transparency still matters.
-

Scented Products: Asset or Risk?
Fragrance can attract or repel customers. Learn how to choose balanced scents, respect scent-free policies, and offer fragrance-free options for wider customer appeal.
-
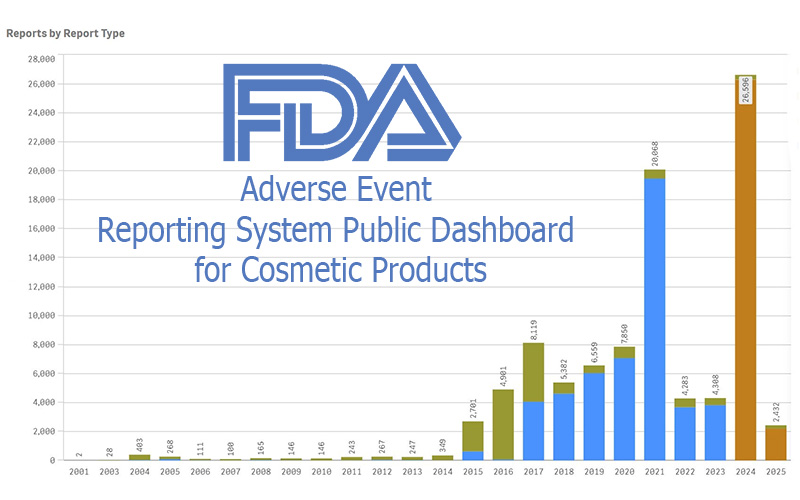
FDA Cosmetics Adverse Event Dashboard
FDA just switched on a live, public cosmetics adverse-event dashboard—big on transparency, light on verification. Here’s how it could help (or hurt) small makers.
-
On the Label: Business Name & Address
Putting your business name and address on the label isn’t just a formality—it’s a legal requirement and a mark of accountability on every product you sell.
-

Label Design: The Physical Label
Your physical label is more than packaging—it’s branding, function, and customer experience combined. Selecting the right material, finish, and adhesive ensures both beauty and durability.
-

MoCRA Rulemaking Updates
The Spring 2025 Unified Agenda, published September 4th, shows more delays in the publication of MoCRA’s fragrance allergen and GMP rules.
-

Product Identity, Product Name, and Brand
Product identity, product name, and brand each serve a different purpose on your label. Here’s how to keep them clear—and compliant.
-

Does AI Give Good Labeling Advice?
I conducted an experiment to determine if AI could help decode cosmetic regulations —but every platform I tested gave some incorrect information. Useful, yes. 100% trustworthy, no.
MORE RESOURCES

Membership
$37 per month
Affordable and easily accessible ongoing support through courses, coaching, regularly scheduled Zoom Q + A sessions, a private Facebook group, and more. You don’t just learn what the rules are—you learn how to apply them to your business and get help when and how you need it.
Label Review
$125 per label
If you want another set of (experienced) eyes on your label to make sure it meets all the requirements, a label review is a good choice. A label review checks your label against the pertinent regulations. You get a checklist showing if corrections are needed (or not), along with documentation to help you understand the requirements. Label reviews take about a week. Once your review is complete, you can send me the revised label and I’ll take a look to make sure everything is in order.
I wanted my labels to be as close to compliant as I could get them. I took advantage of your label review service and can’t thank you enough for your expert advice. It was money well spent! — N.M.
Consulting
$100 – $150 per hour
Consulting is the tailor-made solution to address whatever your concerns are with your labeling, GMP, MoCRA compliance, safety substantiation, and the like. Consulting can be as simple as just answering the questions you have by email, phone or online meeting, or it can cover reviewing all your labels, ingredient declarations, website copy, safety substantiation documentation, and good manufacturing practices. It’s your call – I’m here to help with what you need.
I speak on behalf of the entire team that we are all very appreciative of your work in helping us grow our brand. It is a big step for us, and you have been instrumental in making that a reality. Thank you for your help. — D.C..
Books





